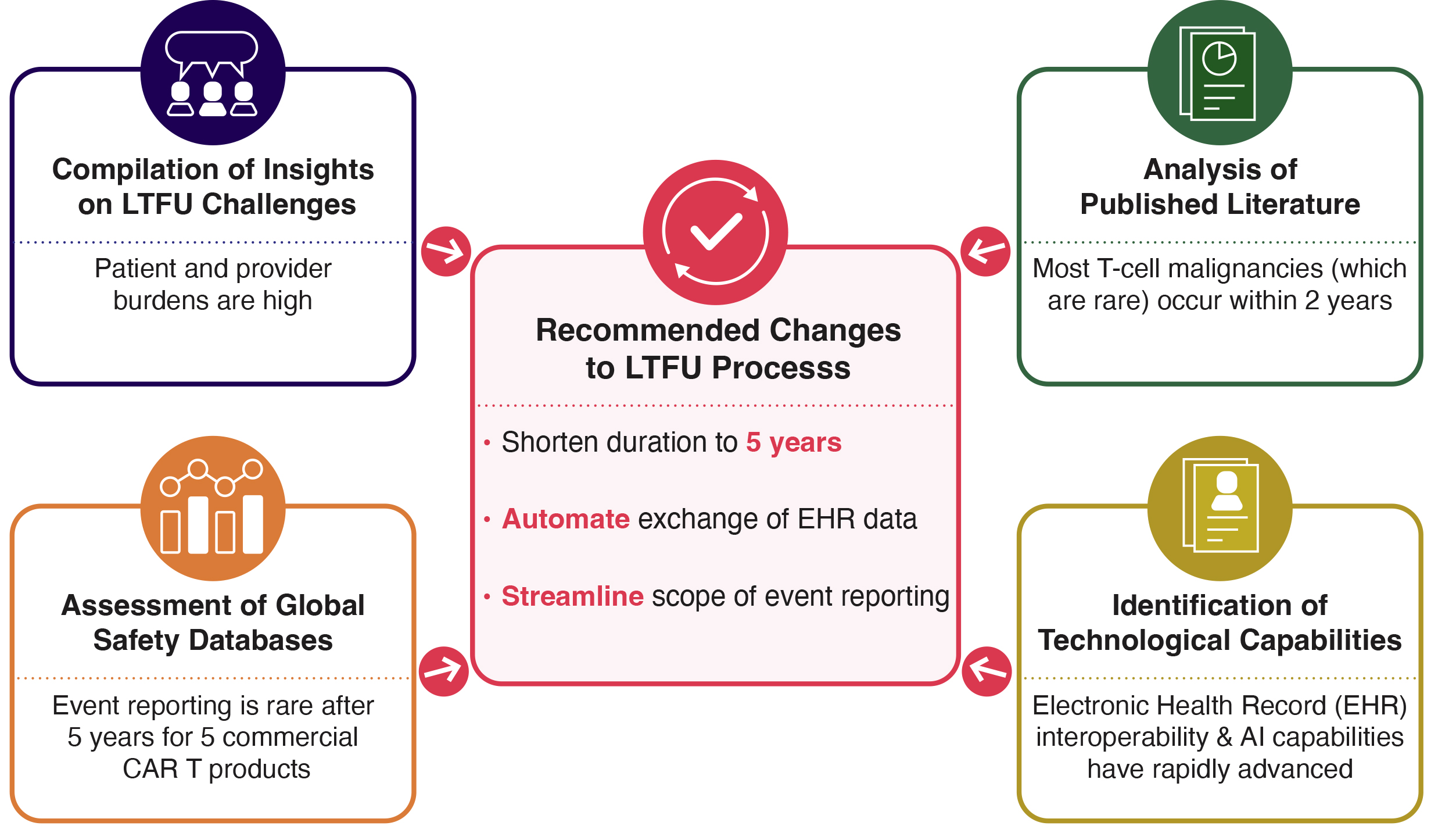
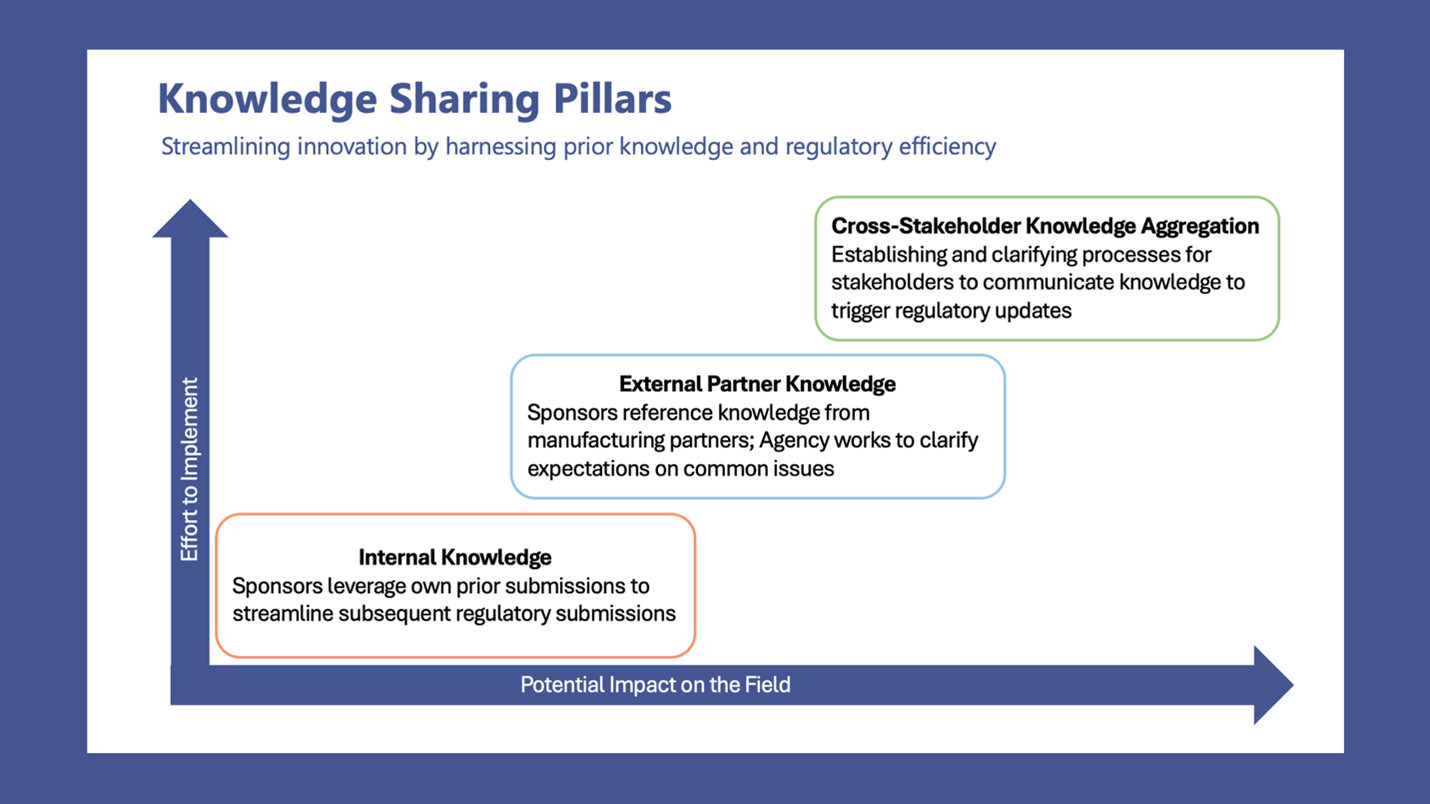
Advancing Pediatric Cell and Gene Therapy Clinical Trials Workshop
March 5, 2026
.jpg)
Workshop | HYBRID
Advancing Pediatric Cell and Gene Therapy Clinical Trials: Scientific, Ethical, Regulatory, and Practical Considerations
Date: April 09, 2026
Time: 9:00 a.m. - 4:30 p.m. ET
Location: Hybrid meeting
For in-person attendees: FDA Headquarters
FDA White Oak Campus
10903 New Hampshire Ave, Building 31
The Great Room
Silver Spring, MD 20903
United States
The FDA’s Center for Biologics Evaluation and Research (CBER), Office of Therapeutic Products (OTP) and the Alliance for Regenerative Medicine (ARM) are co-hosting a hybrid workshop on April 9, 2026, titled “Advancing Pediatric Cell and Gene Therapy Clinical Trials.” The workshop will explore the issues surrounding clinical trials for cell and gene therapies (CGTs) in pediatric populations, particularly for diseases where earlier intervention may result in greater therapeutic benefit. This includes conditions that affect both pediatric and adult populations, as well as diseases that occur primarily in children.
FDA and ARM are convening this public workshop to discuss the following topics with relevant stakeholders:
- Challenges and potential solutions in pediatric clinical trials for CGT products
- Data and information needed to enroll pediatric patients in CGT clinical trials, including interpretations of the prospect of direct benefit
- Appropriate timing to enroll pediatric patients in CGT trials
- Considerations for enrollment of children at presymptomatic and early disease stages
Additional meeting materials and other updates will be posted to this webpage as they become available.
Who should attend:
This workshop is open to the public and is intended for clinicians, scientists, bioethicists, academic and industry representatives, patients, caregivers, and their advocates.
Meeting Logistics and Registration:
This workshop will be held in-person on the FDA White Oak Campus and in a virtual format via YouTube. Registration is required for virtual and in-person attendance. Upon registration, registrants will receive a confirmation email with additional information.
Registration
In-person registration is based on space availability, with priority given to early registrants. Please be prepared to provide complete contact information for each attendee, including name, email address, and affiliation when registering. Registration is required for both in-person and virtual attendance. In-person and virtual registrants will receive an email confirmation after they register with additional workshop details.
To register for the workshop virtually, please use this link:
https://alliancerm.wufoo.com/forms/mdebz3y0eca7uu/
To register for the workshop in-person, please use this link
https://alliancerm.wufoo.com/forms/m17uiwza1fzckdj/
- Registration for in-person attendance will remain open until 11:59 p.m. Eastern Time on Monday, April 6th, 2026. Early registration is recommended because seating is limited; therefore, FDA may limit the number of participants from each organization. If time and space permit, onsite registration on the day of the public meeting (April 9, 2026) will be provided beginning at 9:00 a.m. We will post a notice on the meeting webpage if registration for in-person attendance closes before the day of the public meeting. Campus visitor information will be sent to in-person attendees prior to the workshop.
- Persons attending this conference virtually may register until the start time of the meeting.
Special Accommodations
The FDA will make every effort to accommodate persons with physical disabilities or special needs at public meetings. If you require accommodations due to a disability, email OTPEvents@fda.hhs.gov at least 7 days before the workshop. The FDA White Oak campus and FDA shuttle are generally accessible to people with physical disabilities.
Visiting the FDA Campus
Entrance for meeting attendees (non-FDA employees) is through Building 1. All attendees (FDA and non-FDA) meeting in-person will be required to pass through routine security screening. Laptops, tablets, cell phones, keys, car fobs, coins and other metal items must be removed from pockets and put in a bin or tray for X-ray screening.
Visitors to White Oak campus can use public transportation (Metro train and bus, commuter trains and buses) or taxi and ridesharing services. Limited visitor parking is available. Please allow at least 30 minutes to get from the visitor's parking lots to Building 1 and extra time to accommodate the security check-in. After passing through security screening, attendees will be directed to the workshop room in Building 31.
For additional parking and security information, please refer to Public Meeting Information, Getting to the FDA, and Visitor Parking and Campus Map.
Contact:
For questions regarding visiting the FDA campus, please email:
OTPEvents@fda.hhs.gov
For registration and other questions, please email ARM:
pedscgtctworkshop@catalysthcc.com
Stay Connected
Sign up for the CBER listserv to stay up to date on all FDA CBER news and events.
Alliance for Regenerative Medicine